FDA OneCVM
STACK · Figma · Adobe Creative Cloud · Final Cut Pro · DaVinci Resolve§ 01Context
FDA's Center for Veterinary Medicine reviews submissions affecting the entire U.S. animal-drug supply: pharmaceutical filings, ingredient approvals, labeling, post-market actions. OneCVM is the single portal that ties those reviews together for five distinct audiences: pharmaceutical companies, veterinarians, pet owners, academic researchers, and FDA reviewers.
§ 02The problem
The legacy platform was outdated and inefficient. A single interface was being asked to serve five user populations with non-overlapping needs, vocabularies, and expertise levels, all under strict regulatory compliance. One-size-fits-all design was forcing every audience through a path designed for someone else.
§ 03Approach
- 01
Designed a role-based routing system that lands each audience in a task-specific entry point instead of a shared interface.
- 02
Built a comprehensive design system aligned to FDA branding while introducing modern UX patterns rarely shipped in government software.
- 03
Modernized the platform mascot "Woofie" from a 1980s aesthetic into a contemporary, professional mark.
- 04
Produced wireframes, hi-fi mockups, a demonstration video, and stakeholder presentation materials. Presented directly to FDA program leadership.
§ 04Selected artifacts
Artifacts shown below are placeholders. Cleared, sterilized screens will be supplied for each.
 HI-FI
HI-FI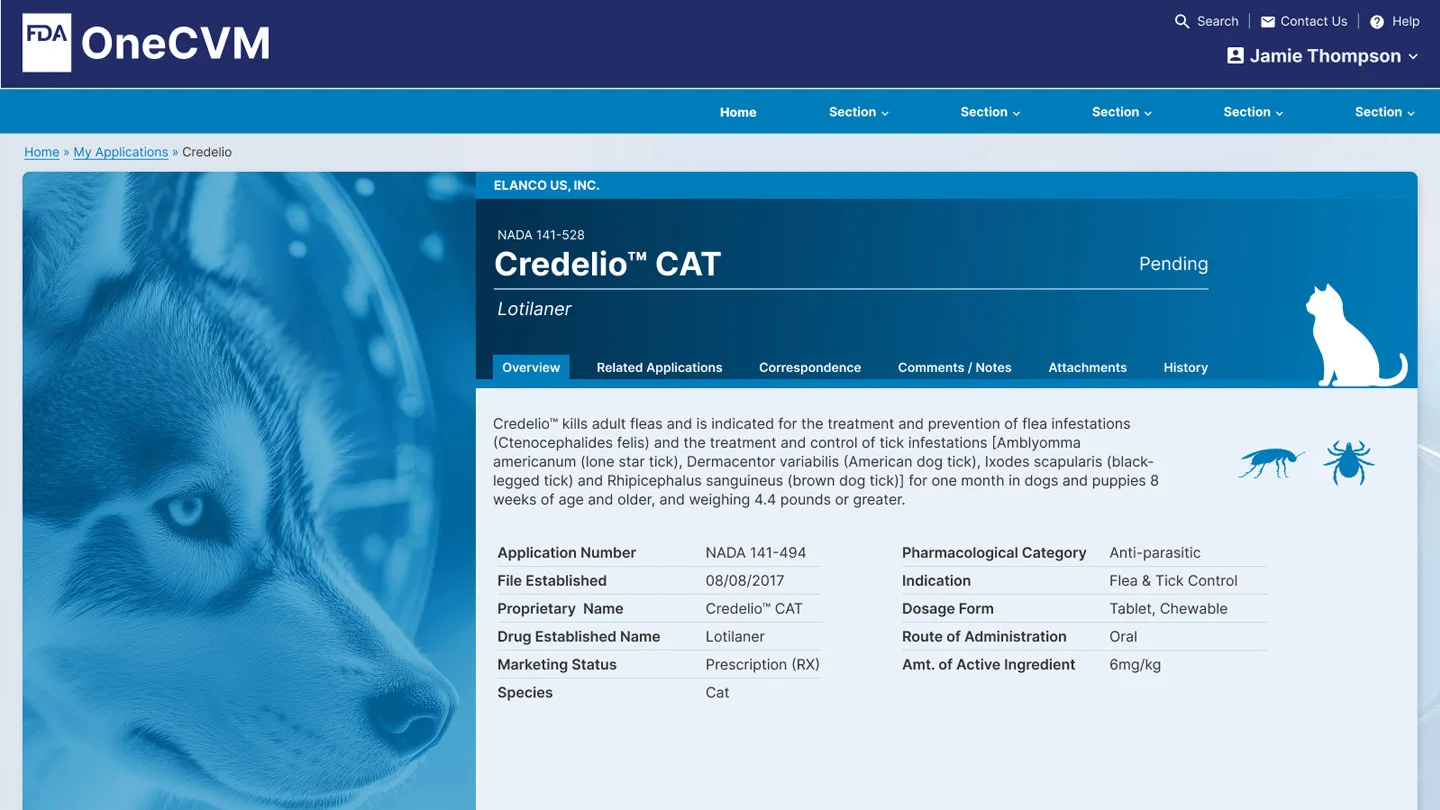 HI-FI
HI-FI MASCOT
MASCOTConcept demonstration approved at 100 percent by FDA senior leadership during the program review. The redesign demonstrated that thoughtful UX could transform complex regulatory systems into accessible, user-centered experiences.
§ 06Reflection
Government systems do not have to look or feel like government systems. Clear information architecture, modern visual design, and intelligent user routing make complex regulatory platforms intuitive without compromising compliance.
